 The free energy change (∆G) of a chemical reaction is a function of its displacement from equilibrium. In biological systems, very often thermodynamically un-favourable energy requiring endergonic reactions couple them to other reactions that liberate free energy (exergonic reactions), so that the overall process is exergonic. Synthesis of ATP during oxidative phosphorylation whose apparent ∆G is as high as 67 kJ mol -1, is also an endergonic process. The syntheses of macromolecules such as proteins and nucleic acids from their simple monomeric components also require input of energy and are therefore, endergonic with +∆G. Conversion of glucose + Pi into glucose-6-phosphate accompanies with a positive free energy change, and thus, is an endergonic reaction. Reactions with a positive ∆G (i.e., ∆G > 0) are known as endergonic reactions or energy consuming reactions. On the other hand, processes with a positive free energy change (+∆G) are not energetically feasible and will not proceed without an input of energy. Gibbs free energy (G) can be defined by combining the enthalpy (H), entropy (S), along with the Kelvin temperature (T) as shown in the following equation, The unit of Gibbs free energy is joules/mole (or calories/ mole). However, a change in Gibbs free energy (∆G) accompanying a process can be measured accurately. Since Gibbs free energy is also a thermodynamic quantity, it is not possible to quantify its absolute value. It is called as Gibbs free energy in honour of Josiah Willard Gibbs (1839-1903), an American mathematician and physical chemist who developed the theory of chemical thermodynamics in 1870s at Yale University and also the concept of free energy. Gibbs Free Energy (G):įree energy is the component of the total energy of a system that is available to do work at constant temperature and pressure and is represented by the symbol G. In all spontaneous reactions such as oxidation of glucose or melting of ice, the ∆S is positive. 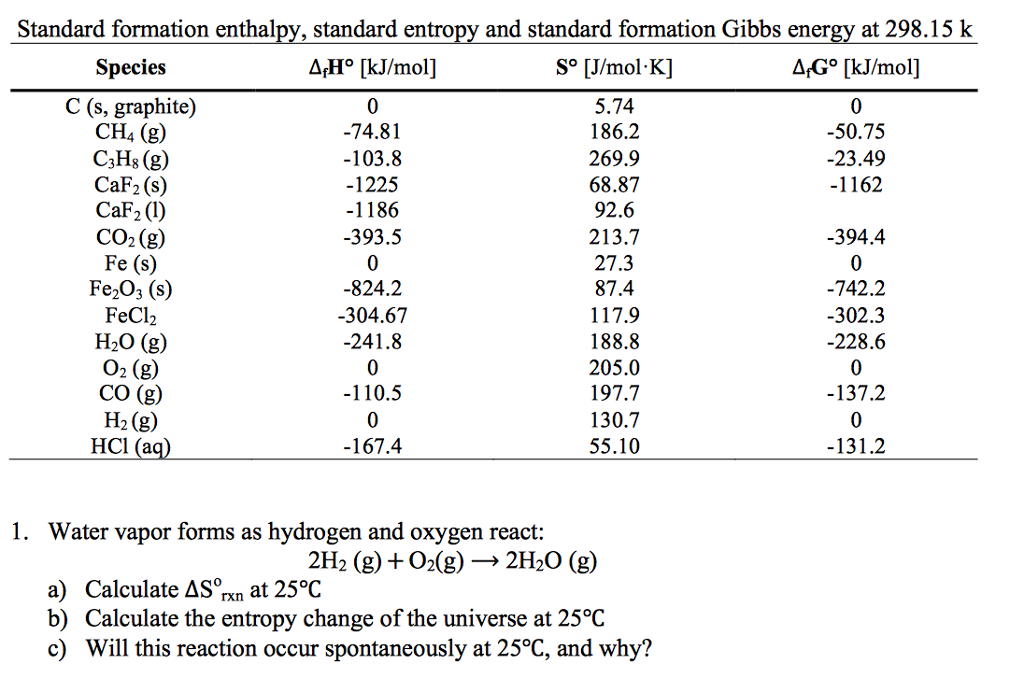
When the products of a reaction are less complex or more disordered than the reactants, the reaction is said to proceed with gain in entropy (+∆S) or vice versa (-∆S). Any change in entropy or disorder accompanying a process from start to finish, is represented by ∆S. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
