 At low concentrations of vinegar, the reaction is limited by the concentration of acid. Use the graph below to answer the following question. vrms3kBTm3 (1.381023J/K)4. What is the mass lost once the Alka Seltzer becomes the limiting reagent The molar mass of NaHCO3 is 84.007 g/mol and the molar mass of carbon dioxide is 44.01 g/mol. The molar mass of carbon dioxide is 44.0095 g/mole. The molar mass of octane is 114.33 g/mole. The mass of a nitrogen molecule is 4.68 1026 kg. Question: Using the following equation for the combustion of octane, calculate the amount of moles of carbon dioxide formed from 100.0 g of octane.  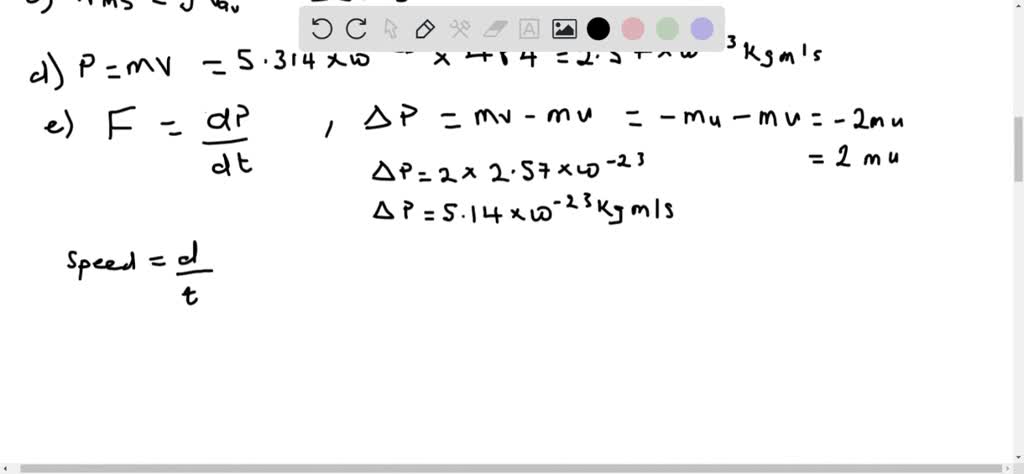
Equation 20.18 can be used to find the rms velocity given the temperature, Boltzmanns constant, and the mass of the atom or molecule. So the molecular weight of Carbon monoxide is 28.0101. The mass of a nitrogen molecule is 4.68 1026 kg. Step 4: Calculate the molecular weight of Carbon monoxide by adding up the total weight of all atoms. Total weight of Oxygen atoms in Carbon monoxide: 15.9994 x 1 = 15.9994 Carbon dioxide is an acidic gas which releases during respiration. Number of Oxygen atoms in Carbon monoxide: 1 Molecular mass is the sum of the masses of all the individual atoms present in a molecule or compound. Finding molar mass starts with units of grams per mole (g/mol). Convert grams Carbon Dioxide to moles - Conversion of Measurement Units. No single carbon atom has a mass of 12.01 amu, but in a handful of C atoms the average mass of the carbon atoms is. Do a quick conversion: 1 grams Carbon Dioxide 0.022722366761722 mole using the molecular weight calculator and the molar mass of CO2. Total weight of Carbon atoms in Carbon monoxide: 12.0107 x 1 = 12.0107 This is the average atomic mass of carbon. Butylene, 56.06 Carbon Dioxide, CO 44.01 Carbon Disulphide, 76.13 Carbon Monoxide. Number of Carbon atoms in Carbon monoxide: 1 Definition and molecular weight (molar mass) of some common substances. Step 3: Calculate the total weight of each atom present in a Carbon monoxide molecule by multiplying its atomic weight by its number. We combine (react) many mers of ethylene. Step 2: Find out atomic weights of each atom (from periodic table).Ītomic weight of Carbon (C): 12.0107 (Ref: Jlab-ele006)Ītomic weight of Oxygen (O) : 15.9994 (Ref: Jlab-ele008) The atomic weight of carbon is 12 and that of hydrogen is 1, so one mer of ethylene has a weight of 2(12) + 4(1) 28. From the chemical formula of Carbon monoxide, you can find that one molecule of Carbon monoxide has one Carbon (C) atom, and one Oxygen (O) atom. Chemical formula of Carbon monoxide is CO. 
Step 1: Find out the chemical formula and determine constituent atoms and their number in a Carbon monoxide molecule.įrom the chemical formula, you will know different atoms and their number in a Carbon monoxide molecule. Calculate the percent yield of carbon dioxide using the actual yield of carbon dioxide in grams from question 2 and the theoretical yield of carbon dioxide in. Note that the value of atomic weight may differ slightly from different sources.ĬALCULATION PROCEDURE: Carbon monoxide Molecular Weight Calculation Make sure you are using the moles of carbon dioxide that can be produced by the limiting reagent that you selected above in part a and the molar mass of carbon dioxide, 44.01 g/mol c. The sum of total weight of all constituent atoms will be the molecular weight of the compound. 
Then calculate the total weight of each atom by multiplying its atomic weight by its number. To calculate molecular weight of any compound, the first step is to know the constituent atoms and their number in that particular compound. In a carbon-dating experiment, a particular type of mass spectrometer is used to separate 14 C ^ ) 2.325 × 1 0 − 26 kg ).The molecular weight of Carbon monoxide is 28.0101. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
